Carbon dots are tiny, fluorescent nanoparticles synthesised from cheap, readily available precursors, but for all their promise in catalysis, sensing, and chemical biology, one question has lingered: what actually makes them good at transferring electrons? This study digs into the surface chemistry of carbon dots to show that amine groups — their number, their density, and how accessible they are — are the decisive factor, and that understanding them quantitatively is the key to designing better nanomaterials.
Our latest work, now published in Small, sheds new light on the structure-reactivity relationships that govern the redox activity of carbon dots (CDs). The study was carried out in collaboration with Prof. Maurizio Prato, Prof. Giacomo Filippini, Prof. Paolo Pengo, and Prof. Jacopo Dosso (University of Trieste, Italy). The research was supported by the European Research Council (ERC AdG-2019 e-DOTS – 885323, ; ERC StG PROTOMAT – 101039578), the University of Trieste, INSTM, the Italian Ministry of Education MIUR (cofin Prot. 2017PBXPN4 and Prot. 20228YFRNL), and by Region FVG Microgrants 2024 (LR 2/2011, Art. 4).
CDs are typically described as core-shell nanoparticles: a rigid carbonaceous core enveloped by a layer of functional groups, among which surface amines are the dominant players, governing solubility, intermolecular interactions, and — as this work clarifies — redox activity. Despite the growing use of CDs across a range of (photo)catalytic applications, a clear, quantitative picture of how surface amine density translates into electron-transfer performance had been missing. To address this, we synthesised and systematically characterised three distinct CD batches prepared from L-arginine alone or in combination with alkyl diamines of different chain lengths, producing a series of nanomaterials with progressively varying amine content and surface accessibility.
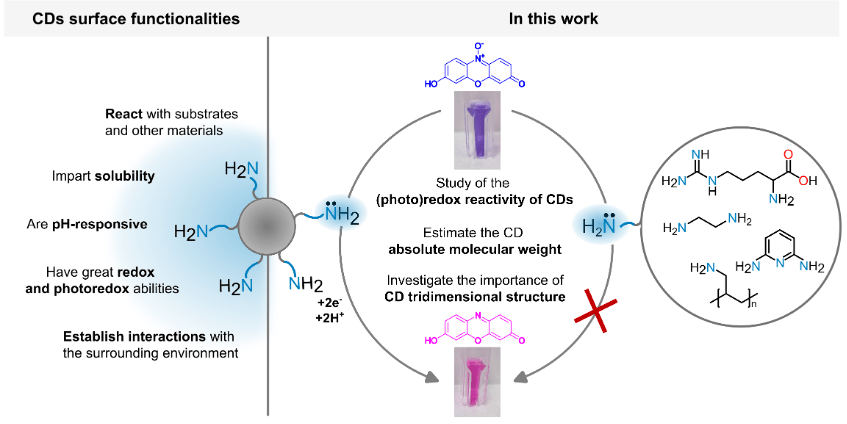
Using the resazurin-to-resorufin reduction as a well-established redox benchmark, we demonstrated that higher amine density leads to markedly superior CD redox performance — both in the dark and under light irradiation. A key mechanistic finding emerged from this analysis: electron transfer does not proceed through isolated amine groups acting independently, but relies on cooperative supramolecular interactions between the CD surface and the substrate, with multiple neighbouring amines working in concert. This cooperative mode of action had not been previously described for this class of materials.
The mechanistic picture was assembled through a multi-technique strategy integrating electrochemical measurements, spectroscopic analyses, and isothermal titration calorimetry. By pairing absolute molecular weight determination — achieved via multi-detection gel permeation chromatography — with thermodynamic binding data, we established a quantitative structure-reactivity framework for rational CD nanoengineering. The outcome is not merely a mechanistic insight, but a practical design blueprint: by tuning amine content and surface arrangement, it becomes possible to engineer CD electron-transfer capabilities with precision, opening the way to next-generation applications in catalysis, materials science, and chemical biology.
Link to the article.


